For a detailed explanation of the general strategy for solving NMR structure elucidation problems, see problem 662.
Here are the steps I would take to solve this problem:
1. Are there any any hints?
Yes. The sharp IR peak at 1,700 cm-1 tells you this molecule contains a carbonyl (C=O).
2. How many IHD are there? (also known as DBE or degrees of unsaturation)
C4H6O2 is the same as C4H6 (oxygens can be ignored) which if fully saturated would be C4H10 (from CnH2n+2).
So this compound is missing 4 H's (C4H10 - C4H6), which corresponds to 2 IHD.
3. Draw some C4H6O2 structures with 2 IHD and eliminate, learn, repeat.
Some things we know so far:
-
From the IR we know this molecule must have a carbonyl group. This "uses" 1C, 1O, and 1 IHD so we have 3C, 1O and 1 IHD remaining to work with).
-
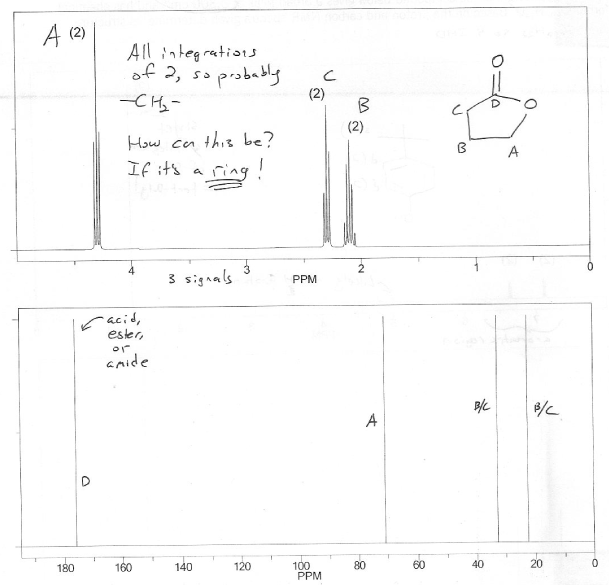
What is the the remaining 1 IHD? It's probably not another carbonyl since the 13C NMR shows only one carbonyl peak (~170 ppm). It's probably not an alkene since we don't see any vinyl hydrogen peaks on the 1H NMR (~5-6 ppm). That leaves a ring.
-
Another clue that this molecule contains a ring is that we only see CH2's in the 1H NMR spectrum (all peaks have an integration of 2).
-
The carbonyl peak on the 13C NMR spectrum is around ~170 ppm, which means it's an oxidation state III carbonyl (ester) rather than an oxidation state II carbonyl (aldehyde or ketone).
Draw a few structures based on these clues and you'll arrive at the correct answer.