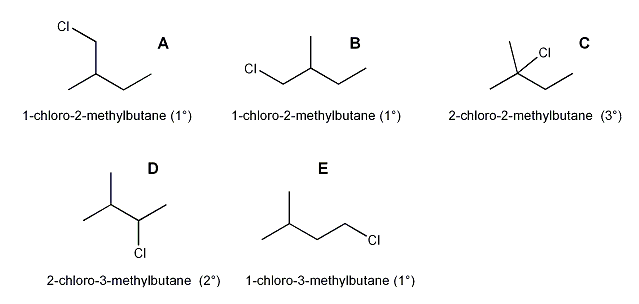
a) There are five different carbons in 2-methylbutane, so hypothetically, you should get five different products, 20% A, 20% B, 20% C, 20% D, and 20% E.
But notice that A and B are the same product! So only four products are possible.
b) Based on statistics alone, we would expect product A (or B) to be the major product, encompassing 40% of all the products (20% A + 20% B).
But this is not what happens in real life because A is primary, and for free-radical halogenation reactions, more substituted products are favored. So the major product will be C, which is tertiary.
This happens because the intermediate is a radical, and radical stability goes 3° > 2° > 1°.
c) Chlorine reacts faster than bromine, and so is less selective than bromine.
For example, we know that the 3° halide will be the major product. But with chlorine, the proportion might turn out to be ~70% 3° , 20% 2°, and 10% 1°.
But since bromine reacts more slowly, you will get better selectivity. The proportion of products will might be something like ~95% 3° , 4% 2°, and 1% 1°.
While on this topic of reactivity, it's interesting to note that iodine reacts too slowly to be useful, and fluorine reacts so violently it's dangerous!