
MS 934 - Carey and Giuliano 8th Ed. CH 19
Solutions can be seen at mendelset.com/ms/934
Description: Goes over carboxylic acid derivatives and the nucleophilic acyl substitution mechanism. Includes synthesis problems.
Total Problems: 6
-
Problem # 726
Show two esters that would yield the two alcohols below after treatment with lithium aluminum hydride.
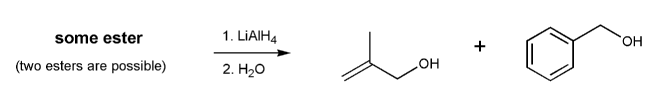
-
Problem # 727
Show how the ester below can be prepared from propene.
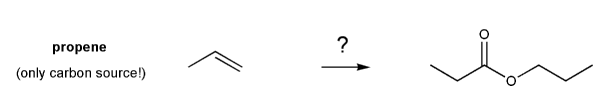
-
Problem # 728
The acyl group is a protecting group for amines. Amines can be acylated using acetic anhydride, and deacylated with base.
Propose a mechanism for each reaction.
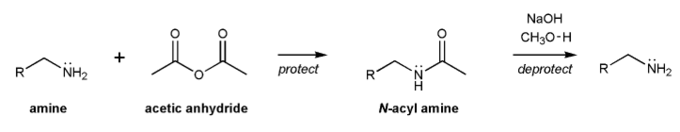
-
Problem # 729
The ester below was dissolved in a solution of water, a small amount of which was isotopically labeled with O-18, denoted with an asterisk.
After a few hours, some isotopically labeled oxygen was found in the ester. Where was it found in the ester? Can you explain why?
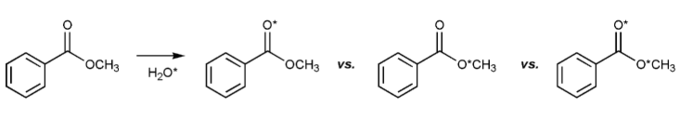
-
Problem # 730
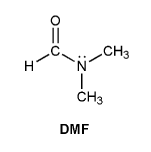
N,N-dimethylformamide (DMF) is shown below. Based on its structure, you might expect to see only one -CH3 signal in the 1H NMR spectrum. But instead DMF shows two different -CH3 signals. Explain.
-
Problem # 735
Show how each ketone below can be prepared from sodium cyanide and either ethylene or propene.
You may also use methyl Grignard and ethylene oxide.
