MS 920 - Aldehydes and Ketones
Solutions can be seen at mendelset.com/ms/920
Description: Problems related to aldehydes and ketones: Wittig, imines, acetals, and protecting groups.
Total Problems: 6
-
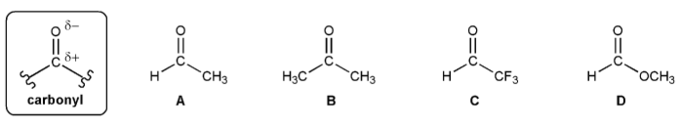
Problem # 710
Rank the carbonyls A-D below in order of decreasing electrophilicity (reactivity with nucleophiles).
(1 = Most reactive). Explain your reasoning.
-
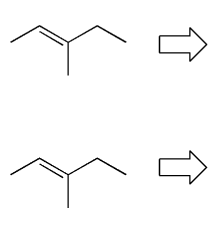
Problem # 711
Show two ways of preparing the alkene below via the Wittig reaction starting from triphenyl phosphine (PPh3).
Is one route better than the other? Why?
-
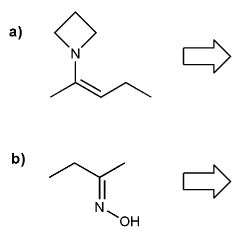
Problem # 712
Show what combination of amine and carbonyl would result in each imine or enamine.
-
Problem # 713
When a carbonyl is treated with semicarbazide under acidic conditions an "imine" is produced called a semicarbazone.
Which of the two products below is the correct structure for a semicarbazone? Explain.
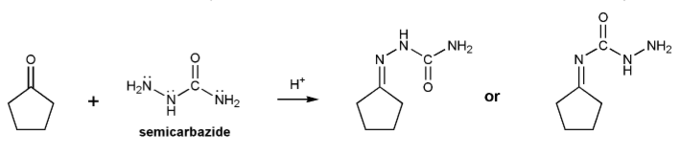
-
Problem # 714
Complete each synthesis below. All carbon sources must come from alkenes.
Each synthesis will involve protecting groups.
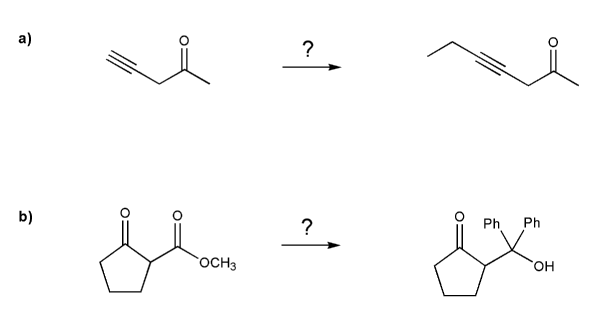
-
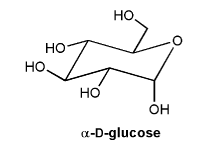
Problem # 715
α-D-glucose is shown below.
a) Is α-D-glucose an acetal, hemiacetal, ketal, or hemiketal?
b) Draw the carbonyl form of α-D-glucose.