MS 917 - Alcohol and Carbonyl Redox
Solutions can be seen at mendelset.com/ms/917
Description: Goes over how alcohols can be oxidized to form aldehydes/ketones and carboxylic acids, which can be transformed further using Grignard reagents, hydride reagents such as NaBH4, and by performing Fischer esterification. Includes several synthesis problems involving carbonyls and epoxides. Also includes an NMR based problem.
Total Problems: 5
-
Problem # 673
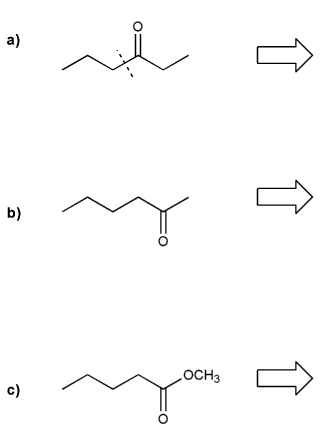
Show how each compound can be prepared from an alkene containing 3 carbons (or less).
Each answer will involve the reaction of a Grignard with either a carbonyl or epoxide.
Note: epoxides are prepared from alkenes using a peroxy acid (epoxidation) such as mCPBA.
-
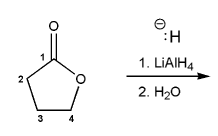
Problem # 674
Show a mechanism for the reduction of butyrolactone using LiAlH4.
-
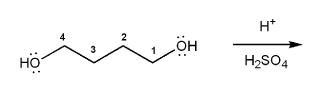
Problem # 677
Show a mechanism for the acid-catalyzed cyclization (condensation) of 1,4-butanediol.
-
Problem # 678
Draw the structure of the major organic product from each reaction sequence.
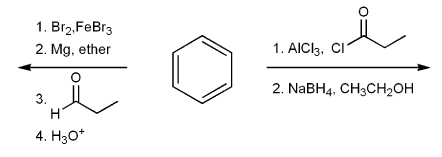
-
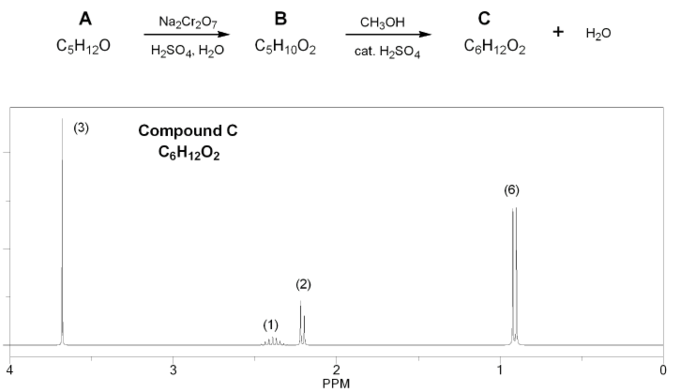
Problem # 679
Compound A (C5H12O) is oxidized using aqueous chromium (Jones reagent) to compound B (C5H10O2), which is then treated with methanol under acidic conditions to yield compound C (C6H12O2) and water.
The 1H NMR of compound C is shown below. Determine the structures of compounds A, B, and C.