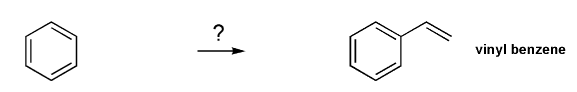MS 913 - Benzene EAS Reactions, EDG/EWG, and ortho/meta/para Directors
Solutions can be seen at mendelset.com/ms/913
Description: Covers electrophilic aromatic substitution (EAS) reactions in detail.
It includes many, many resonance exercises that show why:
- Electron donating groups (EDG) are ortho-para directors
- Electron withdrawing groups (EWG) are meta-directors
- Pyrrole undergoes EAS at C-2 and not C-3
- Pyridine undergoes EAS at C-3 and not C-2 or C-4
- Naphthalene undergoes EAS at C-1 and not C-2
Total Problems: 11
-
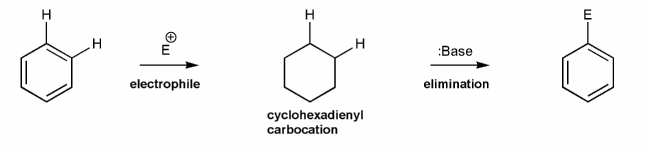
Problem # 587
Use curved arrows to draw a mechanism for the generic electrophilic aromatic substitution (EAS) reaction below.
-
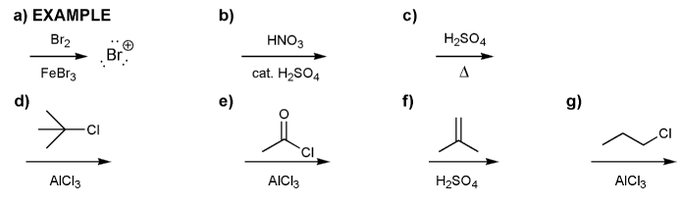
Problem # 596
Indicate the eletrophile formed by each set of reagents/conditions below.
-
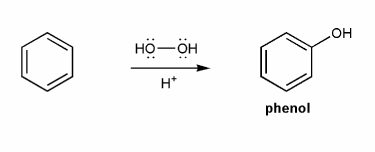
Problem # 595
Phenol can be prepared from benzene and hydrogen peroxide in the presence of a really strong acid. Propose a mechanism for this reaction.
-
Problem # 588
Let's draw resonance forms to see why some groups are EDG or EWG. (I've started you off)
Where are the positive or negative charges placed in EDG/EWG? (ortho/meta/para) Why would this affect EAS reactions?
Note: EDG = electron donating group, EWG = electron withdrawing group
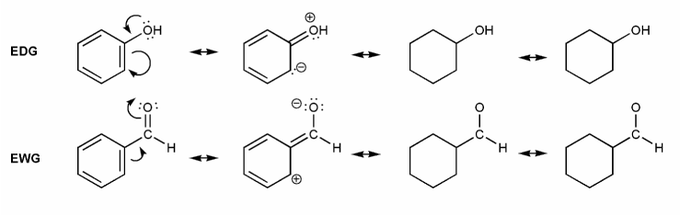
-
Problem # 589
-OR is an EDG and an ortho-para director. Let's draw an EAS reaction's cyclohexadienyl cation intermediates to demonstrate why this is true. I've started you off.
What's good about ortho/para? What's bad about meta?
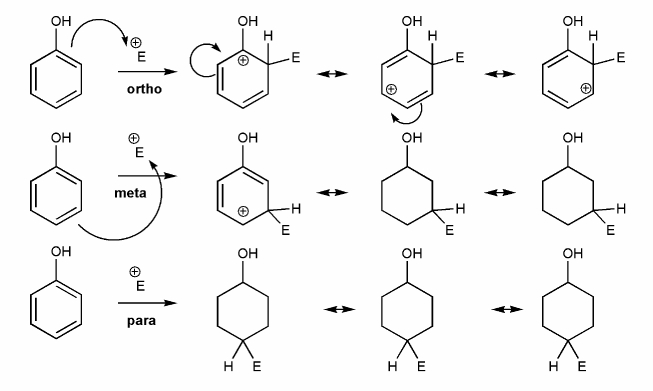
-
Problem # 590
-NO2 is an EWG and a meta director. Let's draw an EAS reaction's cyclohexadienyl cation intermediates to demonstrate why this is true. I've started you off.
What's good about meta? What's bad about ortho/para?
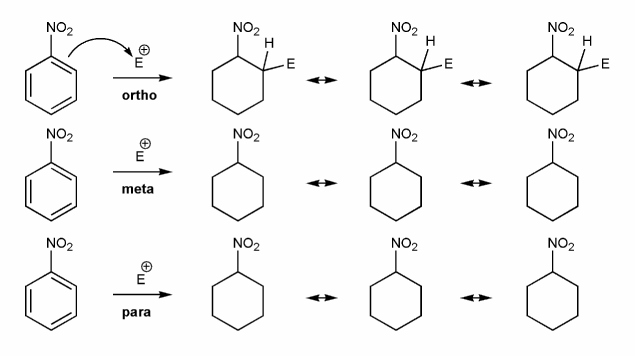
-
Problem # 591
Pyrrole undergoes eletrophilic aromatic substitution at C-2. Let's compare the resonance forms of EAS carbocation intermediates to see why this is the case. What do you think? Why C-2 and not C-3?
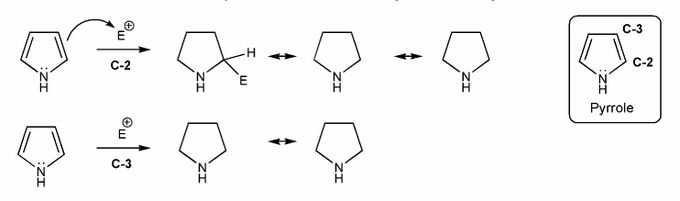
-
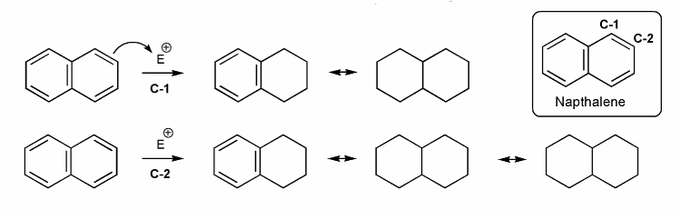
Problem # 592
Naphthalene undergoes eletrophilic substitution at C-1.
Why is this the case, even though substitution at C-2 gives more resonance forms?
-
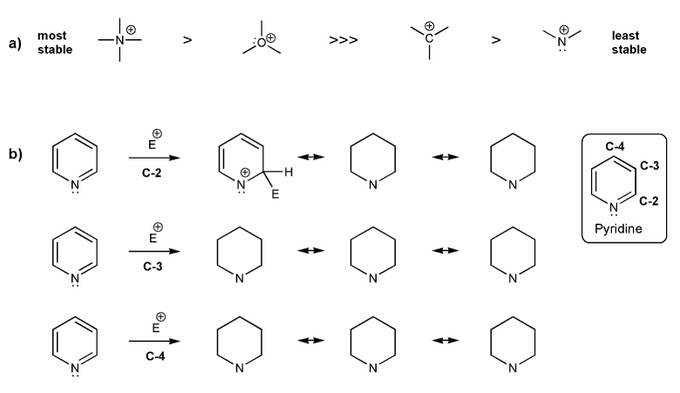
Problem # 593
a) Rationalize the relative stabilities of the cation species below.
b) Pyridine undergoes eletrophilic substitution at C-3. Let's compare the resonance forms of EAS carbocation intermediates to see why this is the case. Consider part a) in your explanation.
-
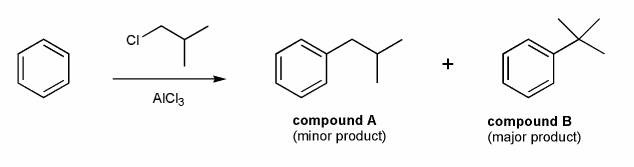
Problem # 594
A chemist tried to prepare compound A from benzene via Friedel-Crafts alkylation and instead produced compound B.
Why did this happen? How could the chemist prepare compound A?
-
Problem # 722
Show how to prepare vinyl benzene from benzene.