MS 906 - Stereochemistry Drills
Solutions can be seen at mendelset.com/ms/906
Description: This mendel set covers various stereochemistry topics, such as:
- Assigning R/S configuration
- Fischer projections
- Identifying meso molecules
- Stereoselective reactions (are the products racemic or optically active?)
- Chiral resolutions
Total Problems: 8
-
Problem # 525
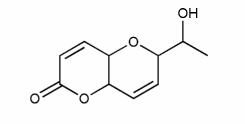
On the molecule below, mark each stereocenter with an asterisk. (Note: in some textbooks, stereocenters are referred to as stereogenic centers, chirality centers, or asymmetric centers).
-
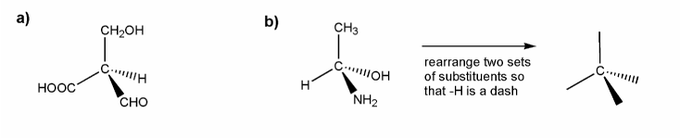
Problem # 526
Assign R or S configuration for each molecule below.
a) is straightforward. I've started you off in b).
-
Problem # 527
Draw the structure of (2R,3S) 2-bromo-3-chlorobutane using wedges and dashes. Also draw a Fischer projection.

-
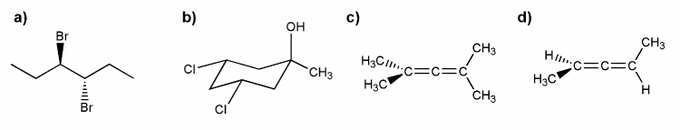
Problem # 528
Indicate which of the molecules below are chiral (if any).
-
Problem # 529
Indicate the major organic product of the reaction below. Include stereochemistry.
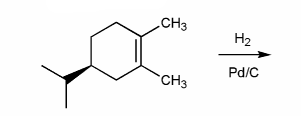
-
Problem # 530Let's work through anti and syn additions to alkenes.Show the product for each reaction below, and indicate whether the product will be a racemic mixture of enantiomers, or a meso compound (which is achiral).
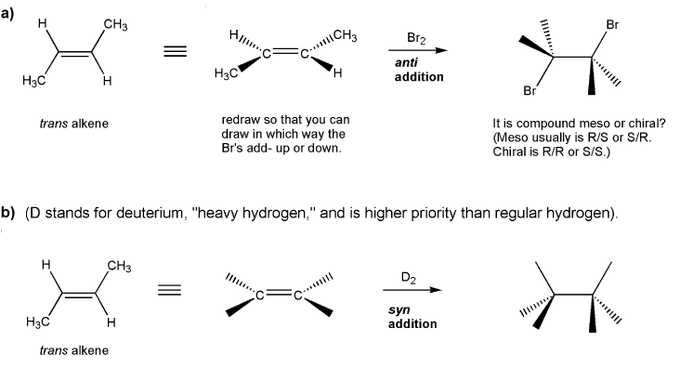
-
Problem # 531E2 elimination reactions require anti-coplanar geometry. (note: some textbooks call this anti-periplanar).Let's work through an E2 reaction, and rotate the molecule eblow into an anti-coplanar geometry to predict the product of this E2 reaction.
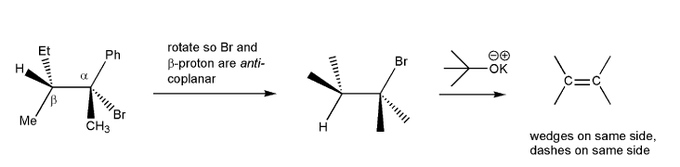
-
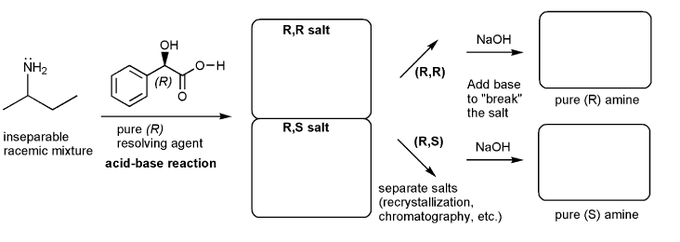
Problem # 532
Let's work through a chiral resolution. Write out the structure of the indicated compound in each box. Include stereochemistry.
Why is it possible to separate the (R,R) and (R,S) salts?