MS 901 - Acid-Base Practice
Solutions can be seen at mendelset.com/ms/901
Description: This mendel set goes through rules that affect acid and base strength:
- Negative charges are best on more electronegative atoms (left to right on periodic table).
- Negative charges are best on larger atoms (up to down on periodic table).
- Resonance stabilizes charges. The more resonance forms the better.
- Neutral compounds are in general more stable than changed ones.
"What makes a compound stable?" problems are key to understand for the MCAT.
Total Problems: 6
-
Problem # 286
Draw the conjugate base forms of each acid listed below, then rank the acids in order or decreasing acidity (1 = most acidic).
Explain your reasoning.
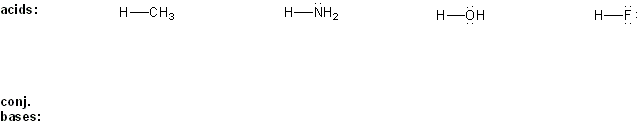
-
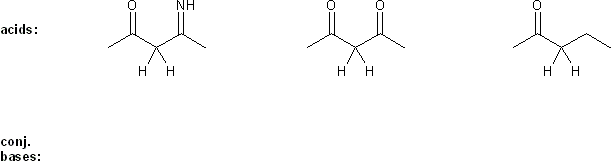
Problem # 288
Draw the conjugate base form of each acid listed below, then rank the acids in order or decreasing acidity (1 = most acidic).
Explain your reasoning.
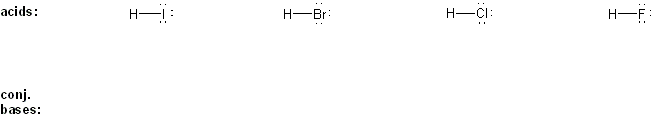
-
Problem # 303
Draw the conjugate base form of each acid listed below, then rank the acids in order or decreasing acidity (1 = most acidic).
Explain your reasoning.
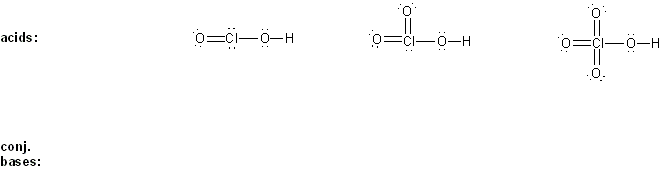
-
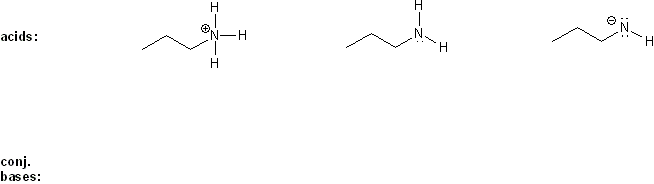
Problem # 304
Draw the conjugate base form of each acid listed below, then rank the acids in order or decreasing acidity (1 = most acidic).
Explain your reasoning.
-
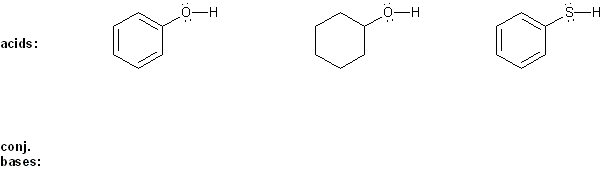
Problem # 305
Rank each group of acids in order of decreasing acidity. (1 = most acidic)
Explain your reasoning. You will have to use more than one rule in your explanation (resonance, electronegativity, atomic radius, etc.).
-
Problem # 306
Rank each group of acids in order of decreasing acidity. (1 = most acidic)
Explain your reasoning. You will have to use more than one rule in your explanation (resonance, electronegativity, atomic radius, etc.).