MS 4638 - Sacramento City College Chem 420 Unit 9 Homework Alkenes (II)
Solutions can be seen at mendelset.com/ms/4638
Description: Please do the following Mendelset problems, and also all of problems #43-69 in Chapter 11 of Second Language.
In addition, please do the odd problems from 70-123 in Chapter 11 of Second Language.
Total Problems: 11
-
Problem # 1336
Determine a synthesis to prepare 2-chloro-4-methylpentane from 1-iodo-4-methylpentane.
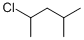 from
from 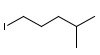
-
Problem # 334
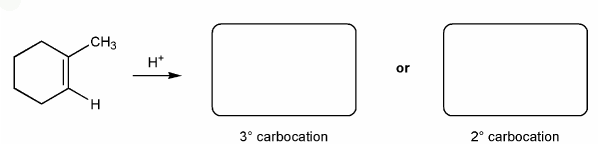
Let's go over how a carbocation can form from an alkene.
Use curved arrows to show the two carbocations that can from from 1-methylcyclohexene.
-
Problem # 336
Predict the product(s) of the reaction below, and used curved arrows to show a mechanism.
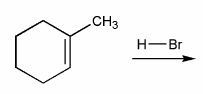
-
Problem # 342
For the reaction below, draw the structures of the carbocation intermediate and the final product.
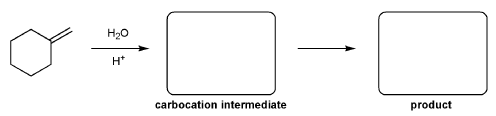
-
Problem # 343
For the reaction below, draw the structures of the chloronium ion intermediate and the final product.
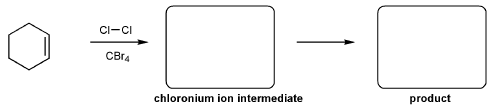
-
Problem # 344
For the reaction below, draw the structures of the borane intermediate and the final product.
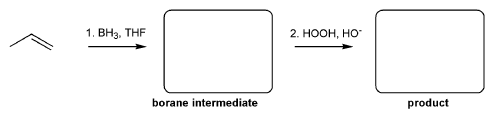
-
Problem # 345
For the reaction below, draw the structures of the radical intermediate and the final product.
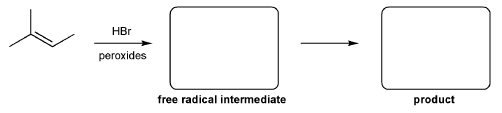
-
Problem # 520
Let's work through an alkene addition reaction. Draw the structures for each of the species in the three boxes below (3º carbocation, protonated thiol, and thiol). Also draw curved arrows to show electron movement. Note: thiol = RSH, like an alcohol, but with sulfur instead of oxygen.
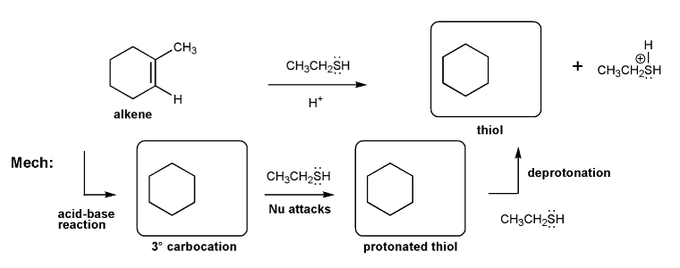
-
Problem # 521
Let's work through a halogenation reaction. Draw the structures for each of the species in the four boxes below (3º carbocation, halonium ion, protonated thiol, and thiol). Also draw curved arrows to show electron movement. Note: thiol = RSH, like an alcohol, but with sulfur instead of oxygen.
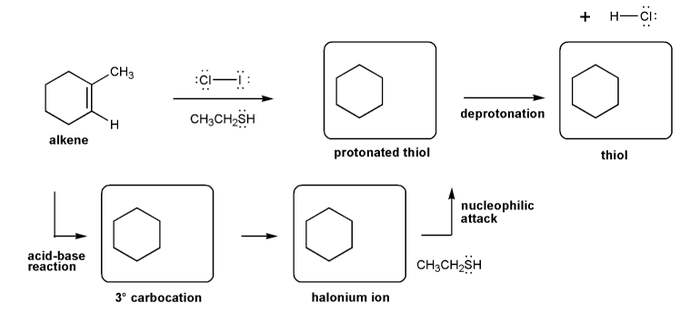
-
Problem # 529
Indicate the major organic product of the reaction below. Include stereochemistry.
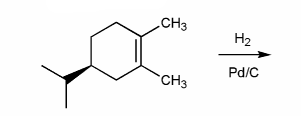
-
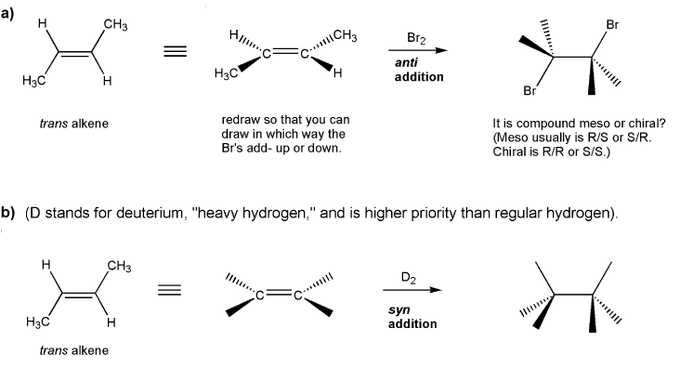
Problem # 530Let's work through anti and syn additions to alkenes.Show the product for each reaction below, and indicate whether the product will be a racemic mixture of enantiomers, or a meso compound (which is achiral).