
MS 4608 - SCC Chem 420 Dr. Zenner Unit 7 Homework Set
Solutions can be seen at mendelset.com/ms/4608
Description: Unit 7 Homework: Please complete the EVEN problems in Chapters 9 and 10 of your Second Language text, and the following problems from Mendelset.
Total Problems: 15
-
Problem # 660
Using your knowledge of 1H NMR, predict the NMR spectrum for the compound below. (draw out the spectrum you would expect to see). Be sure to include:
- peak integrations
- peak multiplicities
- chemical shifts (approximate)
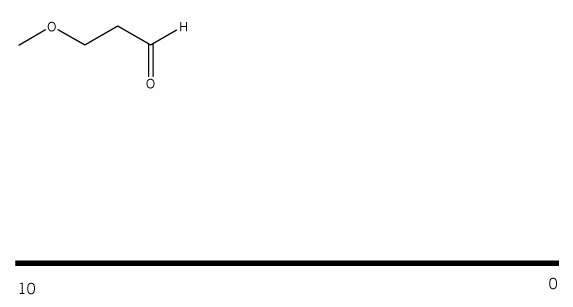
-
Problem # 662
The 1H and 13C NMR spectra of a compound with chemical formula C10H14O are shown below. The compound's IR spectrum shows a broad peak at 3,300 cm-1. Determine the structure of this compound.
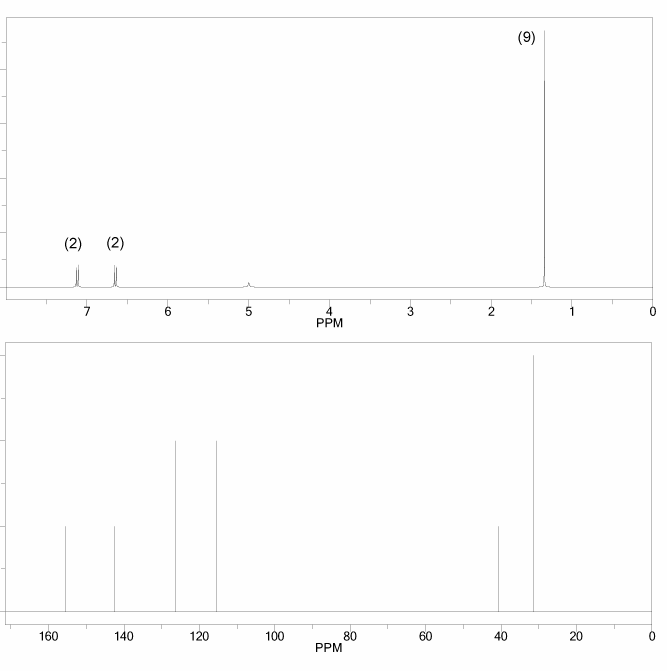
-
Problem # 663
The 1H and 13C NMR spectra of a compound with chemical formula C4H6O2 are shown below. The compound's IR spectrum shows a sharp peak at 1,700 cm-1. Determine the structure of this compound.
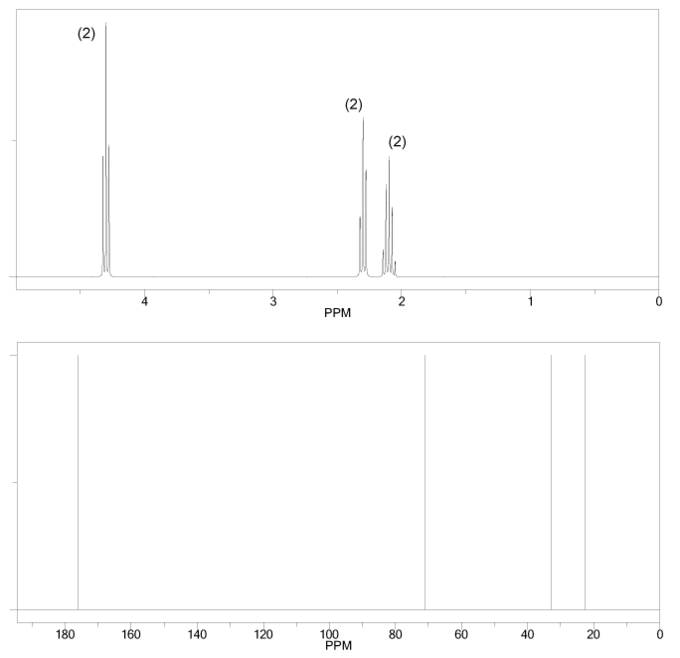
-
Problem # 534
Using curved arrows, draw the mechanism for the SN2 reaction below.
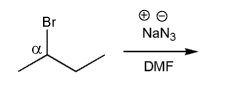
-
Problem # 348
For the reaction below, draw the structures of the carbocation intermediate and the final product.
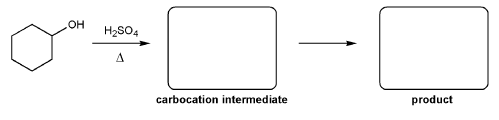
-
Problem # 535
Rank the following anions in order of decreasing stability (1 = most stable)
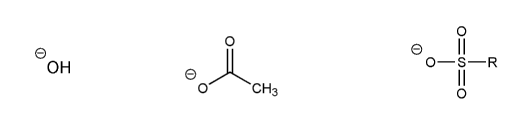
-
Problem # 536
Rank the following electrophiles in order of decreasing reactivity with NaN3 in DMF. (1 = most reactive)
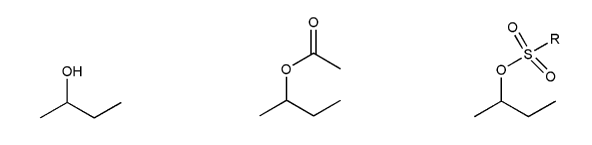
-
Problem # 537
Indicate the reagents necessary to carry out each transformation.
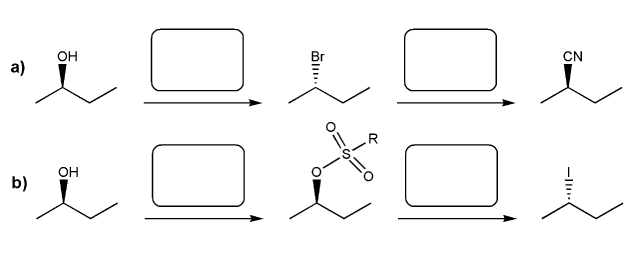
-
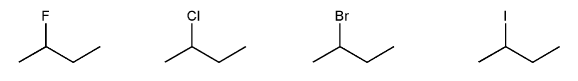
Problem # 538
Rank the following electrophiles in order of decreasing reactivity with NaN3 in DMF. (1 = most reactive)
-
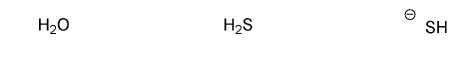
Problem # 539
Rank the following compounds in order of decreasing nucleophilicity. (1 = most nucleophilic)
-
Problem # 540
Rank the following anions in order of decreasing stability (1 = most stable)

-
Problem # 541
Rank the following compounds in order of decreasing reactivity with NaI in acetone. (1 = most reactive)
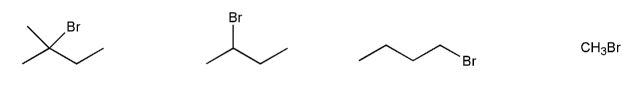
-
Problem # 542
Rank the following compounds in order of decreasing reactivity with water (solvolysis). (1 = most reactive)
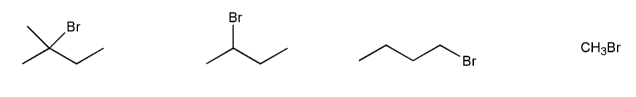
-
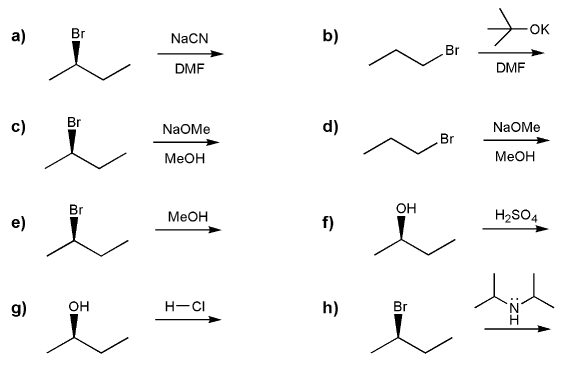
Problem # 560
For each reaction below, determine whether the primary reaction is SN1, SN2, E1, or E2, and then draw the product.
Note: Me = methyl (CH3)
-
Problem # 703
Show two ways to prepare the ether below from a combination of an alcohol and an alkyl halide via the Williamson ether synthesis.
Is one way better than the other? Why?
