MS 4553 - SCC Chem 420 Unit 4 Homework Set
Solutions can be seen at mendelset.com/ms/4553
Description: Please complete ALL the problems in Ch 4 of "Second Language", and the ODD problems in Chapters 5 and 6 of "Second Language", as well as the following problem set.
Total Problems: 12
-
Problem # 320
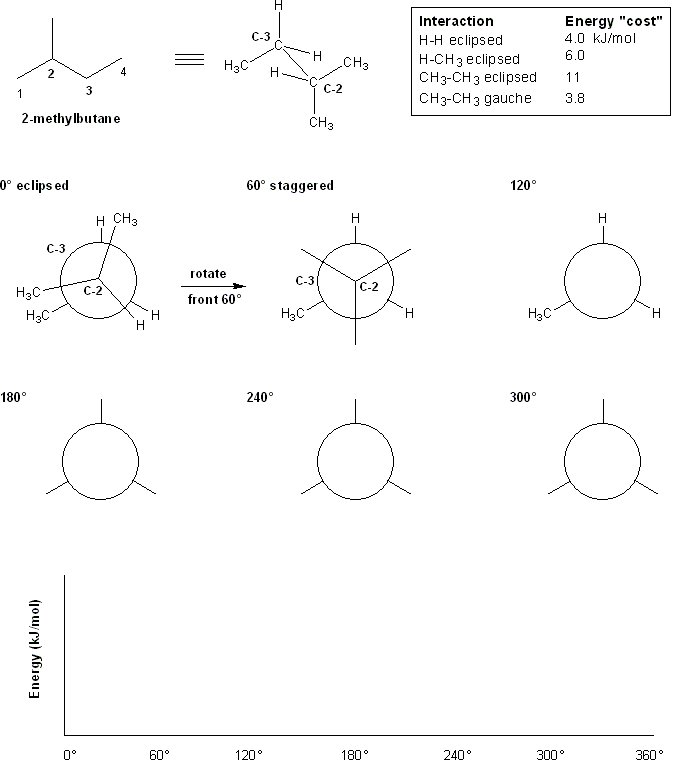
Let's perform conformational analysis on 2-methylbutane along the C2-C3 bond. We'll use the energy chart given below.
First, draw out the Newman projections along the C2-C3 bond, rotating the front carbon (C-2) by 60 degrees clockwise each time while keeping the back carbon (C-3) stationary.
According to the table above, how much energy does each conformation "cost?"
Second, make a plot of the total energy value for each Newman projection versus its dihedral angle.
-
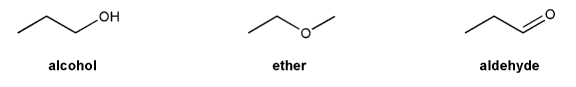
Problem # 697
Rank the following compounds in order of decreasing boiling point.
Also, make a guess about their relative solubilities in water. Explain your reasoning.
-
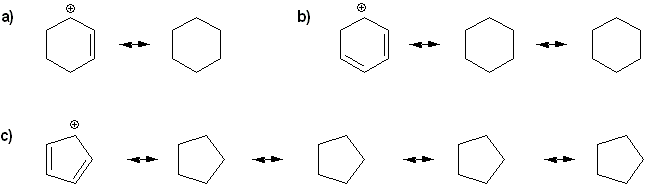
Problem # 315
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
-
Problem # 309
Convert each formula to a carbon skeleton diagram, or vice-versa.
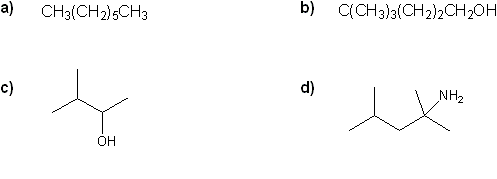
-
Problem # 310
For each molecule, determine the formal charge of the indicated atom.
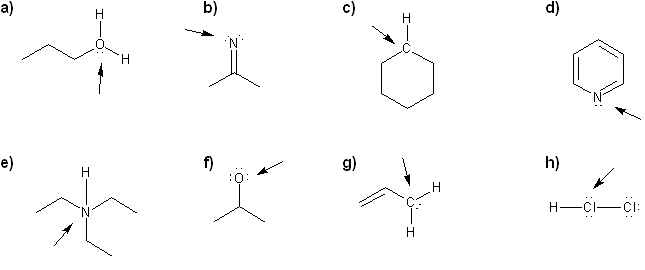
-
Problem # 311
For each molecule below, draw in all implied lone pairs and/or protons (hydrogens) based on the formal charge shown.
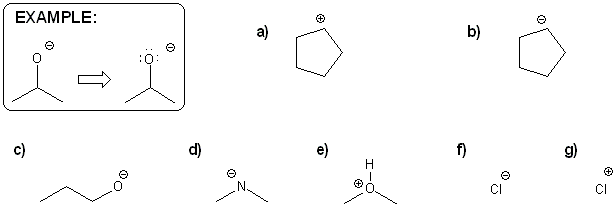
-
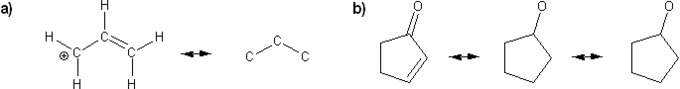
Problem # 312
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
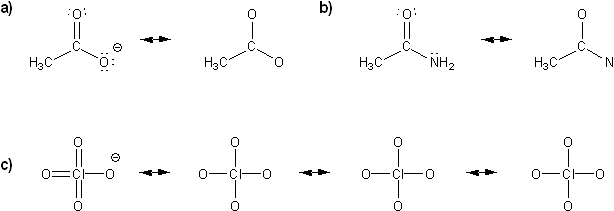
-
Problem # 313
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
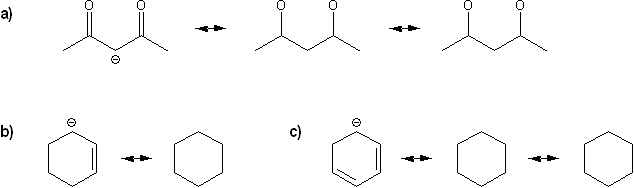
-
Problem # 314
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
-
Problem # 1287
Rank each set of compounds in order of decreasing boiling point (1 = highest boiling point):
a) ethane, n-octane, n-pentane
b) n-butane, 1-butanol, 1-chlorobutane.
c) n-octane, 2-methylheptane, 2,5-dimethylhexane
(Note that the n- prefix before an alkane just means that it's one chain, without any branching.)
-
Problem # 317
α-D-Glucose is shown below. Draw its two chair forms. Which conformation is more stable? Explain.
I recommend using the common convention wedge = "up" and dash = "down."
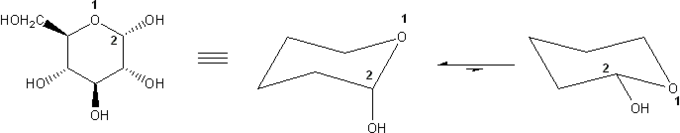
-
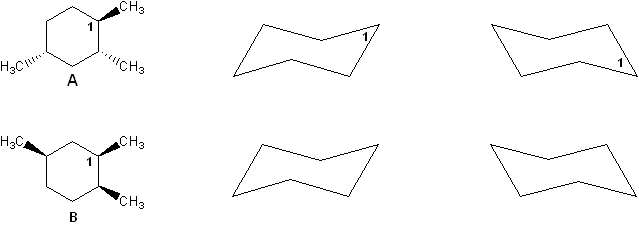
Problem # 318
Two stereoisomers of trimethylcyclohexane are shown below (compounds A and B). Compare cyclohexane chair forms to determine which isomer has a lower heat of combustion. Explain your reasoning.