MS 4526 - Chem 420 Unit 3 Homework
Solutions can be seen at mendelset.com/ms/4526
Description: Please Read Chapter 3 of "2nd Language" and do the associated problems.
In addition, complete the following problem set.
Total Problems: 10
-
Problem # 308
Rank the group of molecules below in in order of decreasing basicity. (1 = most basic)
Explain your reasoning.
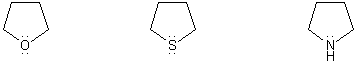
-
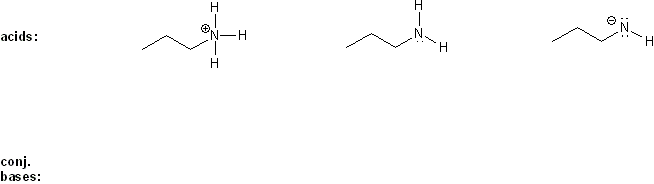
Problem # 304
Draw the conjugate base form of each acid listed below, then rank the acids in order or decreasing acidity (1 = most acidic).
Explain your reasoning.
-
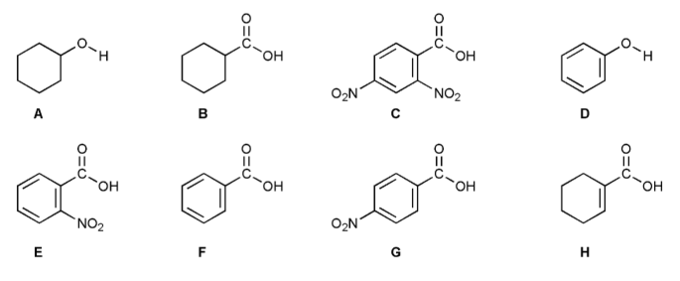
Problem # 717
Rank each of the eight compounds A through H below in order of decreasing acidity (1 = most acidic).
Don't get intimidated! What are the differences between these compounds?
Consider electron withdrawing groups (EWG), resonance, hybridization, and the functional group of the acidic proton.
-
Problem # 721
Rank each of the four compounds below in order of decreasing acidity (1 = most acidic).
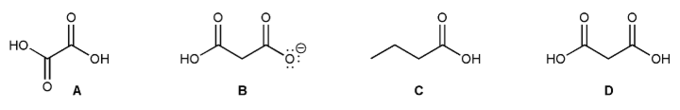
-
Problem # 753
Rank the amines W through Z below in order of decreasing basicity (1 = most basic). Explain your reasoning.
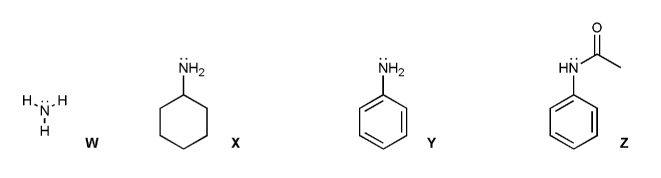
-
Problem # 306
Rank each group of acids in order of decreasing acidity. (1 = most acidic)
Explain your reasoning. You will have to use more than one rule in your explanation (resonance, electronegativity, atomic radius, etc.).
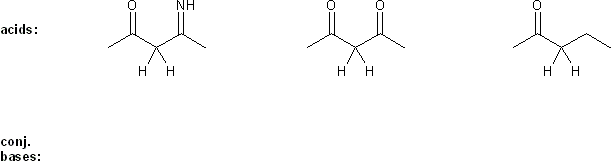
-
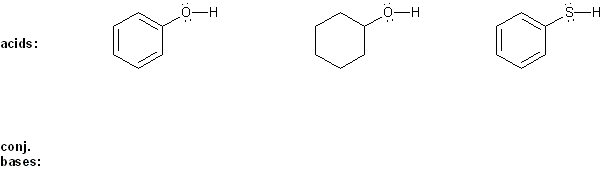
Problem # 303
Draw the conjugate base form of each acid listed below, then rank the acids in order or decreasing acidity (1 = most acidic).
Explain your reasoning.
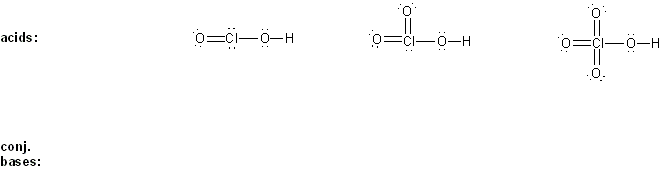
-
Problem # 720
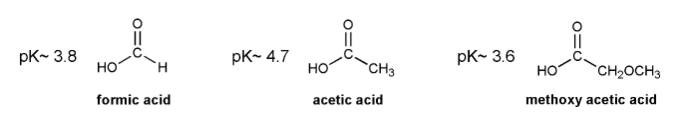
Base your answers to the three problems below on your knowledge of electron donating groups and electron withdrawing groups (EDG and EWG).
a) Based on the pKa's listed below, Is formic acid more or less acidic than acetic acid? Propose an explanation why.
b) If acetic acid were added to a pH = 4.7 buffer solution, what percentage of it would be in its acetate (conjugate base) form?
c) Methoxy (-OCH3) is usually considered an EDG. But based on the pKa of methoxy acetic acid, do you think this is always the case? Explain.
-
Problem # 305
Rank each group of acids in order of decreasing acidity. (1 = most acidic)
Explain your reasoning. You will have to use more than one rule in your explanation (resonance, electronegativity, atomic radius, etc.).
-
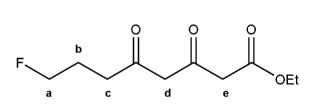
Problem # 745
The molecule below has five different types of hydrogens (A through E). Rank each in order of decreasing acidity.
(1 = most acidic). Explain your reasoning.