MS 2693 - mendal
Solutions can be seen at mendelset.com/ms/2693
Description: my problem
Total Problems: 13
-
Problem # 312
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
-
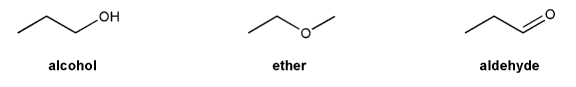
Problem # 313
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
-
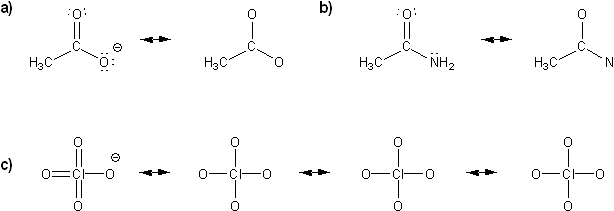
Problem # 311
For each molecule below, draw in all implied lone pairs and/or protons (hydrogens) based on the formal charge shown.
-
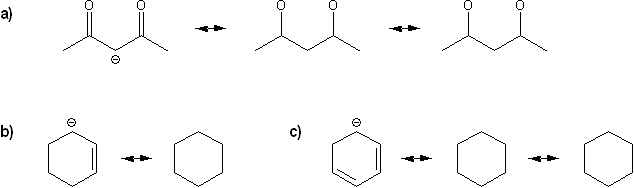
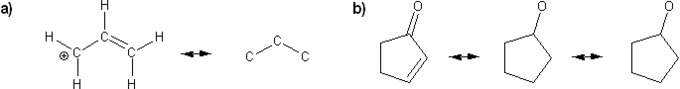
Problem # 314
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
-
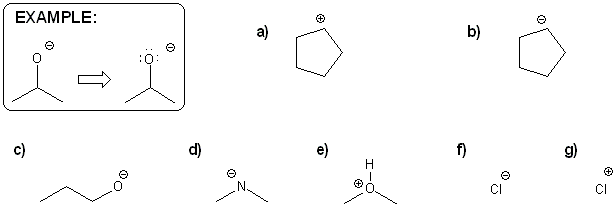
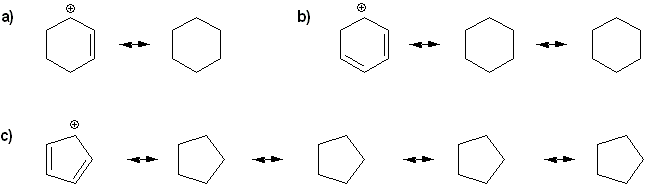
Problem # 315
Draw all possible resonance forms for each structure below. Use curved arrows.
Note that some structures only show charge, and not implied protons or lone pairs!
-
Problem # 309
Convert each formula to a carbon skeleton diagram, or vice-versa.
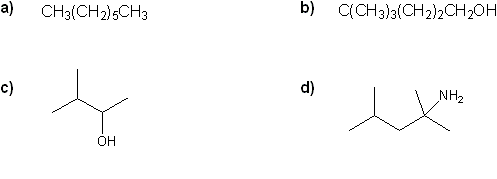
-
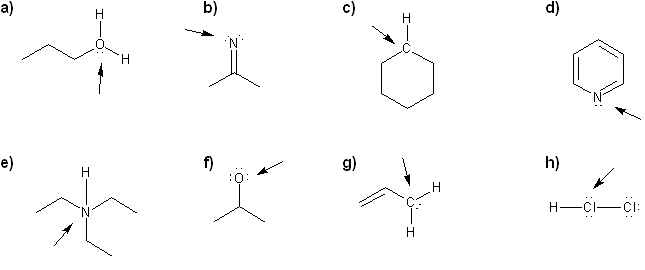
Problem # 310
For each molecule, determine the formal charge of the indicated atom.
-
Problem # 1287
Rank each set of compounds in order of decreasing boiling point (1 = highest boiling point):
a) ethane, n-octane, n-pentane
b) n-butane, 1-butanol, 1-chlorobutane.
c) n-octane, 2-methylheptane, 2,5-dimethylhexane
(Note that the n- prefix before an alkane just means that it's one chain, without any branching.)
-
Problem # 569
Draw all resonance forms for each species.
For the anion and cation species, used curved arrows. For the radical species, use hooks.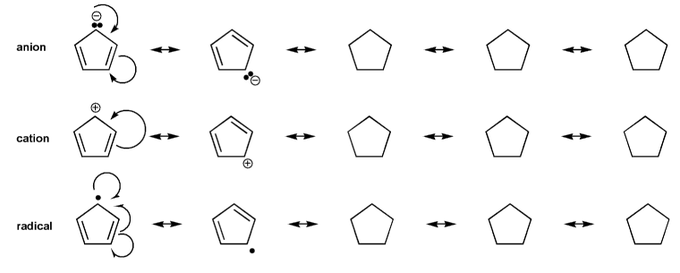
-
Problem # 580
Draw all resonance forms for each species.
For the anion and cation species, used curved arrows. For the radical species, use hooks.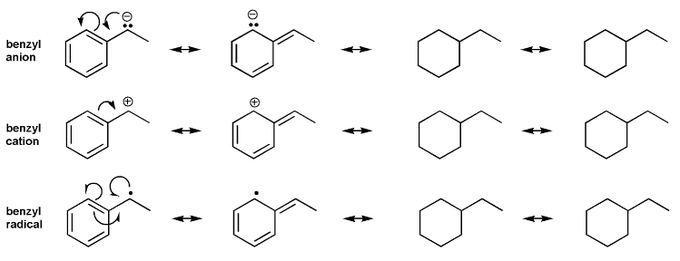
-
Problem # 755
The nitrosyl cation is shown below. Also shown are several proposed resonance arrows, only one of which is correct.
Draw the resonance forms that would follow from each set of arrows, and include formal charges. Which one is the correct resonance form? Explain your reasoning.
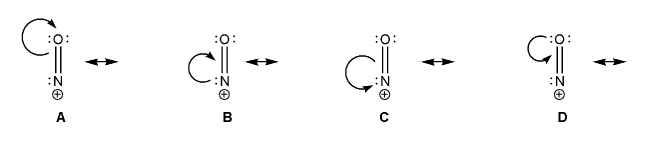
-
Problem # 1286
For the molecule shown below, indicate the hybridization (sp3, sp2, sp, etc.) of atoms A through H, and the bond angles of X, Y, and Z.
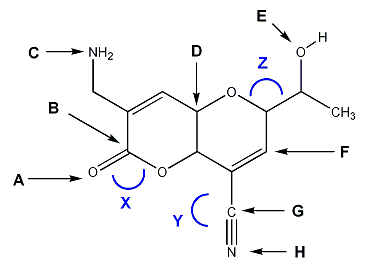
-
Problem # 697
Rank the following compounds in order of decreasing boiling point.
Also, make a guess about their relative solubilities in water. Explain your reasoning.