MS 1317 - Practice 1
Solutions can be seen at mendelset.com/ms/1317
Description: Practice 1
Total Problems: 10
-
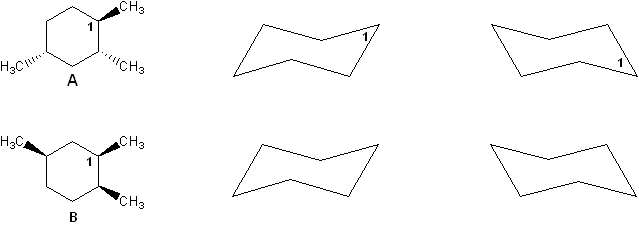
Problem # 318
Two stereoisomers of trimethylcyclohexane are shown below (compounds A and B). Compare cyclohexane chair forms to determine which isomer has a lower heat of combustion. Explain your reasoning.
-
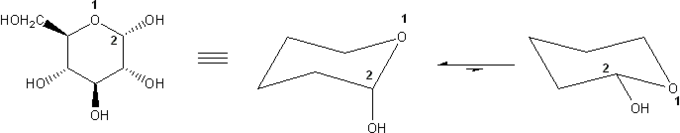
Problem # 317
α-D-Glucose is shown below. Draw its two chair forms. Which conformation is more stable? Explain.
I recommend using the common convention wedge = "up" and dash = "down."
-
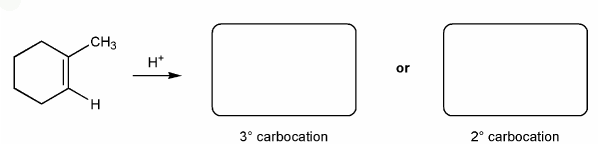
Problem # 334
Let's go over how a carbocation can form from an alkene.
Use curved arrows to show the two carbocations that can from from 1-methylcyclohexene.
-
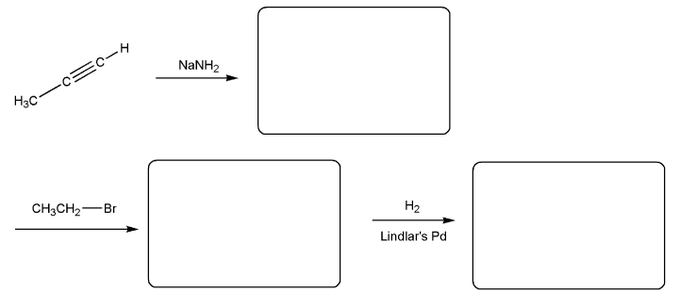
Problem # 336
Predict the product(s) of the reaction below, and used curved arrows to show a mechanism.
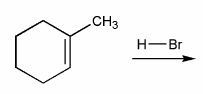
-
Problem # 342
For the reaction below, draw the structures of the carbocation intermediate and the final product.
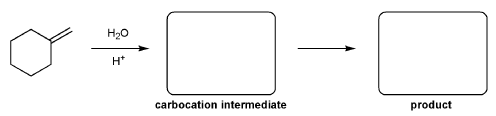
-
Problem # 343
For the reaction below, draw the structures of the chloronium ion intermediate and the final product.
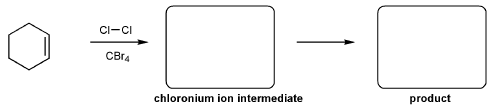
-
Problem # 344
For the reaction below, draw the structures of the borane intermediate and the final product.
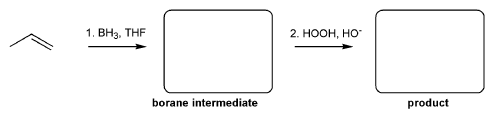
-
Problem # 520
Let's work through an alkene addition reaction. Draw the structures for each of the species in the three boxes below (3º carbocation, protonated thiol, and thiol). Also draw curved arrows to show electron movement. Note: thiol = RSH, like an alcohol, but with sulfur instead of oxygen.
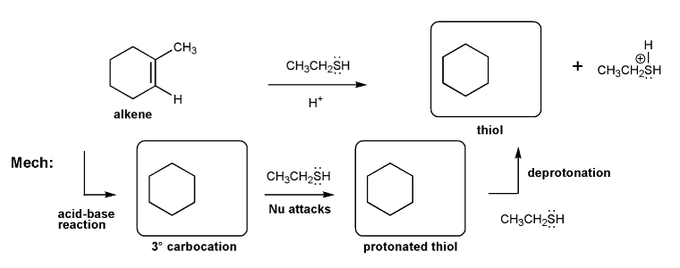
-
Problem # 529
Indicate the major organic product of the reaction below. Include stereochemistry.
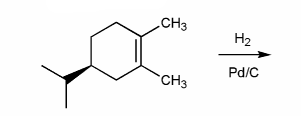
-
Problem # 561
Fill in the product for each reaction below. Indicate stereochemistry where appropriate.