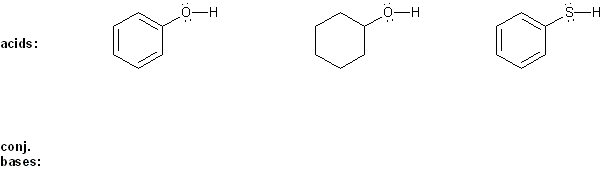
Rank each group of acids in order of decreasing acidity. (1 = most acidic)
Explain your reasoning. You will have to use more than one rule in your explanation (resonance, electronegativity, atomic radius, etc.).
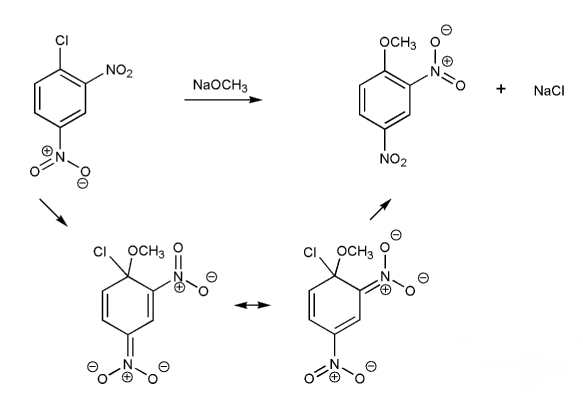
Draw in the arrows to show the electron flow and resonance forms in the nucleophilic aromatic substitution reaction below.
Note: Depending on the textbook, nucleophilic aromatic substitution is referred to as NAS, SNAr, or addition-elimination.
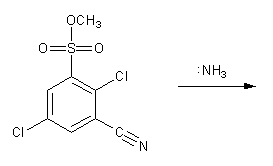
Draw a mechanism for the nucleophilic aromatic substitution (SNAr) reaction below. Show all resonance forms of the intermediate.
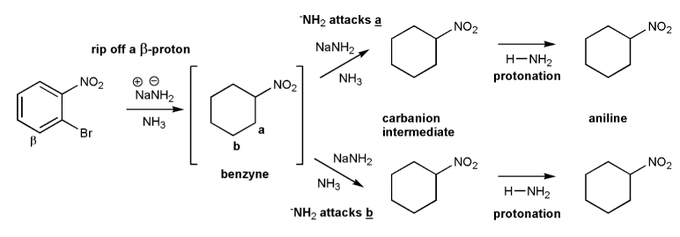
Let's go through a benzyne reaction (also called elimination-addition).
In the reaction below, the strong base (NaNH2) will form a benzyne intermediate, which when forms either ortho nitroaniline or meta nitroaniline.
Used curved arrows to show the formation of each intermediate and the final products.
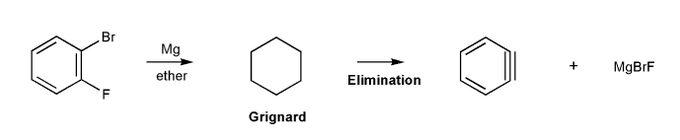
Let's go through another way to make benzyne.
First, let's form a Grignard reagent. Then, let's elminate to form benzyne.