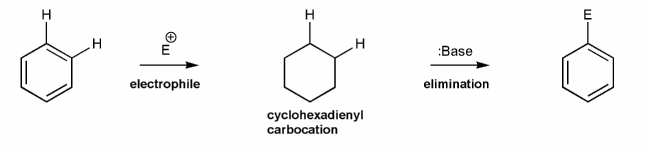
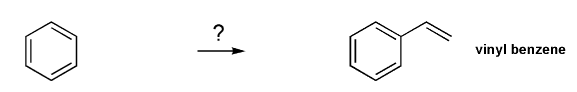Use curved arrows to draw a mechanism for the generic electrophilic aromatic substitution (EAS) reaction below.
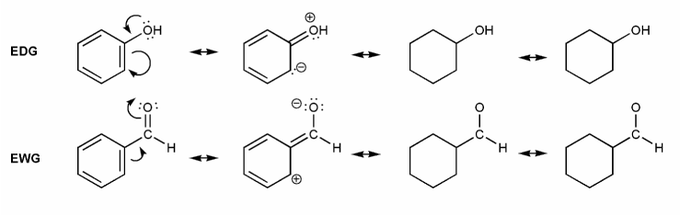
Let's draw resonance forms to see why some groups are EDG or EWG. (I've started you off)
Where are the positive or negative charges placed in EDG/EWG? (ortho/meta/para) Why would this affect EAS reactions?
Note: EDG = electron donating group, EWG = electron withdrawing group
-OR is an EDG and an ortho-para director. Let's draw an EAS reaction's cyclohexadienyl cation intermediates to demonstrate why this is true. I've started you off.
What's good about ortho/para? What's bad about meta?
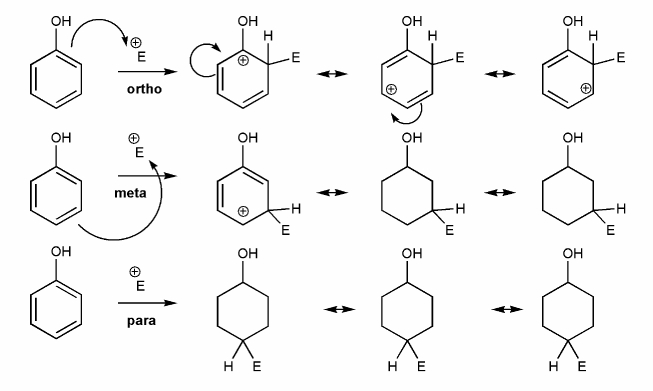
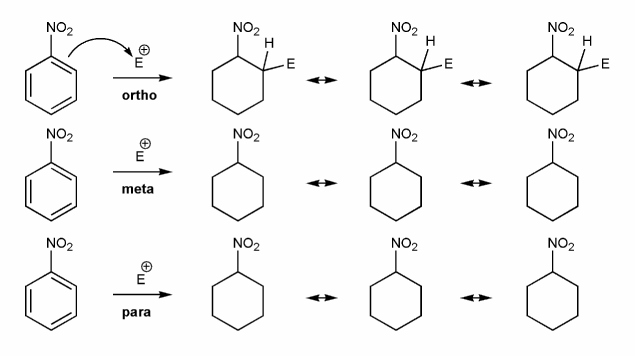
-NO2 is an EWG and a meta director. Let's draw an EAS reaction's cyclohexadienyl cation intermediates to demonstrate why this is true. I've started you off.
What's good about meta? What's bad about ortho/para?
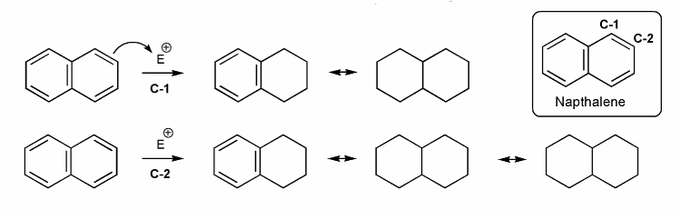
Naphthalene undergoes eletrophilic substitution at C-1.
Why is this the case, even though substitution at C-2 gives more resonance forms?
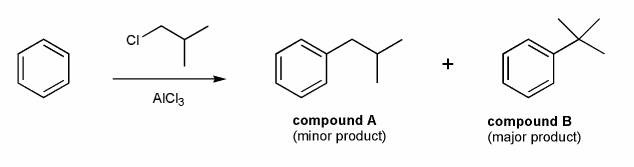
A chemist tried to prepare compound A from benzene via Friedel-Crafts alkylation and instead produced compound B.
Why did this happen? How could the chemist prepare compound A?
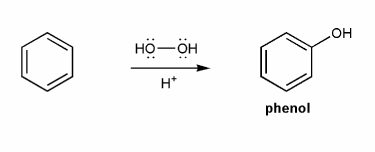
Phenol can be prepared from benzene and hydrogen peroxide in the presence of a really strong acid. Propose a mechanism for this reaction.
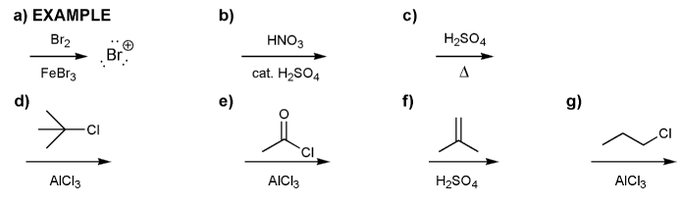
Indicate the eletrophile formed by each set of reagents/conditions below.
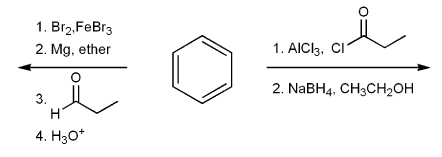
Draw the structure of the major organic product from each reaction sequence.
Show how to prepare vinyl benzene from benzene.