

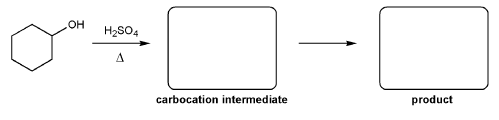
For the reaction below, draw the structures of the carbocation intermediate and the final product.
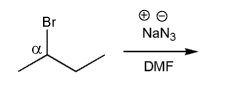
Using curved arrows, draw the mechanism for the SN2 reaction below.
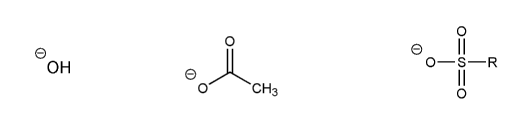
Rank the following anions in order of decreasing stability (1 = most stable)
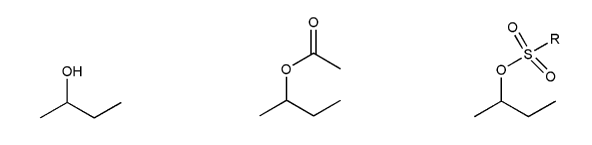
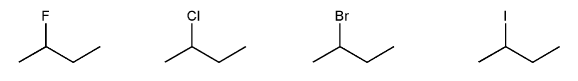
Rank the following electrophiles in order of decreasing reactivity with NaN3 in DMF. (1 = most reactive)
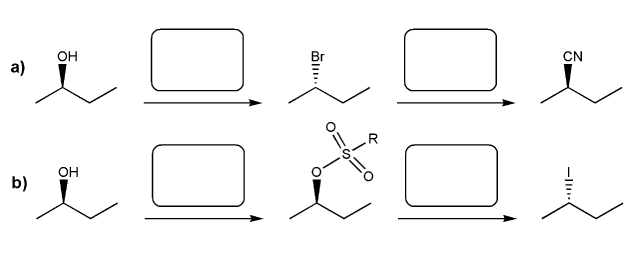
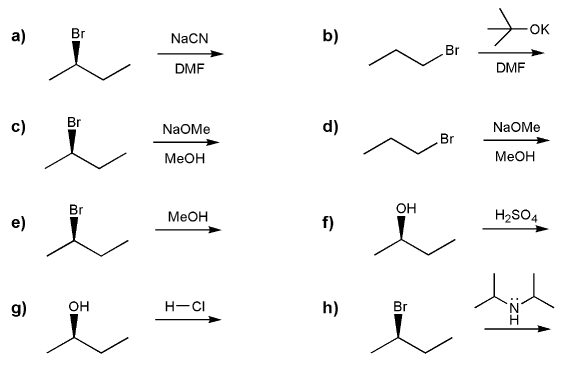
Indicate the reagents necessary to carry out each transformation.
Rank the following electrophiles in order of decreasing reactivity with NaN3 in DMF. (1 = most reactive)
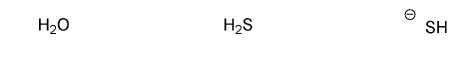
Rank the following compounds in order of decreasing nucleophilicity. (1 = most nucleophilic)
Rank the following anions in order of decreasing stability (1 = most stable)

Rank the following compounds in order of decreasing reactivity with NaI in acetone. (1 = most reactive)
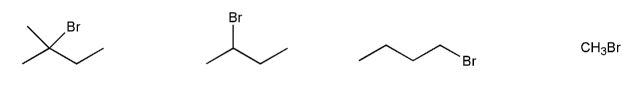
Rank the following compounds in order of decreasing reactivity with water (solvolysis). (1 = most reactive)
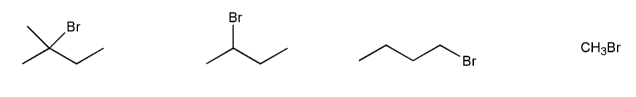
For each reaction below, determine whether the primary reaction is SN1, SN2, E1, or E2, and then draw the product.
Note: Me = methyl (CH3)
Show two ways to prepare the ether below from a combination of an alcohol and an alkyl halide via the Williamson ether synthesis.
Is one way better than the other? Why?
